Carbonate To Bicarbonate
The carbonate ion is the simplest oxocarbon anion. When the first proton is donated HCO3- otherwise known.

Multimedia Forming A Precipitate Chapter 6 Lesson 3 Chemical Equation Middle School Chemistry Lesson
So in a weird way by increasing our pH to become hydronium deficient equilibrium shifts towards the intuitively lower pH side of the reaction to try and counteract this and we get carbonate as.

Carbonate to bicarbonate. When atmospheric carbon dioxide is dissolved in seawater carbonic acid H 2 CO 3 is formed. The compound will readily absorb. It has a molecular mass of 6001 gmol and carries a total formal charge of 2.
Of its transformation into bicarbonate and carbonate in a slightly alkaline aqueous medium and they contain about 60 times as much inorganic carbon as is in the atmosphere. K CaCO Ca CO o CaCO 3 2 - 3 2 3 19. Recipe can be automatically scaled by entering desired final volume.
Tests for carbonate ion compounds reactions precipitates. The molecular weight of carbonate is 60 grams and bicarbonate is 61 grams. This regulates your bodys pH or.
- sometimes called supercarbonate. Calcium Carbonate is used as the source of CO2Carbon dioxide and the resultant calcium oxide is used to recover the ammonia from. Answer 1 of 2.
The carbonate ion is the anion of carbonic acid which is a weak acidSome compounds of carbonate ion forms precipitates and some metal carbonates are soluble in waterAlso some metal carbonates have colours in. We can quantify the carbonate CO3 and bicarbonate HCO3 concentration and as well as hydro Oxley OH in the water form the analyzed parameter of Alkalinity P. Carbonate and Bicarbonate The carbon dioxide that is dissolved by naturally circulating waters appears in chemical analysis principally as bicarbonate and carbonate ions.
Calcium carbonate in water with a fixed partial pressure of carbon dioxide. NaHCO3 Sodium Bicarbonate is mainly prepared by the Solvay Process which is the reaction of sodium chloride ammonia and carbon dioxide in water. Carbonate has a -2 charge while bicarbonate has a -1 charge due to the extra hydrogen proton it contains.
Bicarbonate is excreted and reabsorbed by your kidneys. A carbonate in which but half the hydrogen of the acid is replaced by a positive element or radical thus making the proportion of the acid to the positive or basic portion twice what it is in the normal carbonates. They convert it.
Your blood brings bicarbonate to your lungs and then it is exhaled as carbon dioxide. When we deal with brewing water chemistry we are actually working with equivalents which is equal to the weight of the molecule divided by its electrical charge. Carbonate-Bicarbonate Buffer pH 92 to 106 preparation guide and recipe.
Carbon dioxide and water will be given off leaving dry sodium carbonate. Can be divided into various species if inflection points are known a. The CarbonateBicarbonate Buffer System.
Your kidneys also help regulate bicarbonate. Calculation of Bicarbonate Carbonate and Hydroxide Alkalinity 1. However ecient transfer takes place only into the topmost 100 m wind-mixed layer.
For every one mole of hydronium relative to bicarbonate we have two moles of hydronium relative to carbonate. Carbonate-bicarbonate buffer is used extensively in molecular and cell. Bicarbonate is an important substrate that contributes to various metabolic reactions such as the biosynthesis of arginine uracil or fatty acids or to the degradation of toxic cyanate Aguilera Van Dijken De Winde.
This is the soda ash. Remember total alkalinity is the amount of acid required to lower the pH of the solution to 45 2. For a given pH the concentration of each species can be computed multiplying the respective alpha by the concentration of total calcium carbonate originally present.
Phenolphthalein alkalinity hydroxide and ½ carbonate neutralized occurs when. Carbon dioxide plays a vital role in the chemistry of sea water. Carbonic acid is diprotic which means in has two H ions to donate to solution.
The large supply of atmospheric carbon dioxide is partly intercepted by photosynthesizing vegetation. For the case of a fixed partial pressure of carbon dioxide and calcium carbonate dissolved in the aqueous phase one more equation is need to describe the system. It consists of one carbon atom surrounded by three oxygen atoms in a trigonal planar arrangement with D3h molecular symmetry.
This is the solubility product of calcium carbonate. It is the conjugate base of the hydrogen carbonate bicarbonate ion HCO. The chemical reaction for the process is.
Carbonate that follows this path represents a linkage between the carbon cycle and the hydrologic cycle. Bicarbonate and carbonate are converted to carbon dioxide by lowering the pH of the solution then the absorbance of the dissolved carbon dioxide at 2345 wavenumbers is measured using a liquid sample cell. Simply heat baking soda or sodium bicarbonate in a 200 F oven for about an hour.
Bicarbonate also known as HCO3 is a byproduct of your bodys metabolism. Commonly used for various immunoassay applications and for many protein and antibody conjugation procedures including sandwich ELISA which require experimental surface coatings. Advances in Genetics 2014.
A bit over 6 bicarbonate ion takes over and reigns up to pH a bit over 10 from where fully ionized carbonate ion takes over. Elleuche Pöggeler 2008a.

Sodium Bicarbonate Iupac Name Sodium Hydrogen Carbonate Commonly Known As Baking Soda Is A Chemical Compo Chemistry Science Chemistry Sodium Bicarbonate
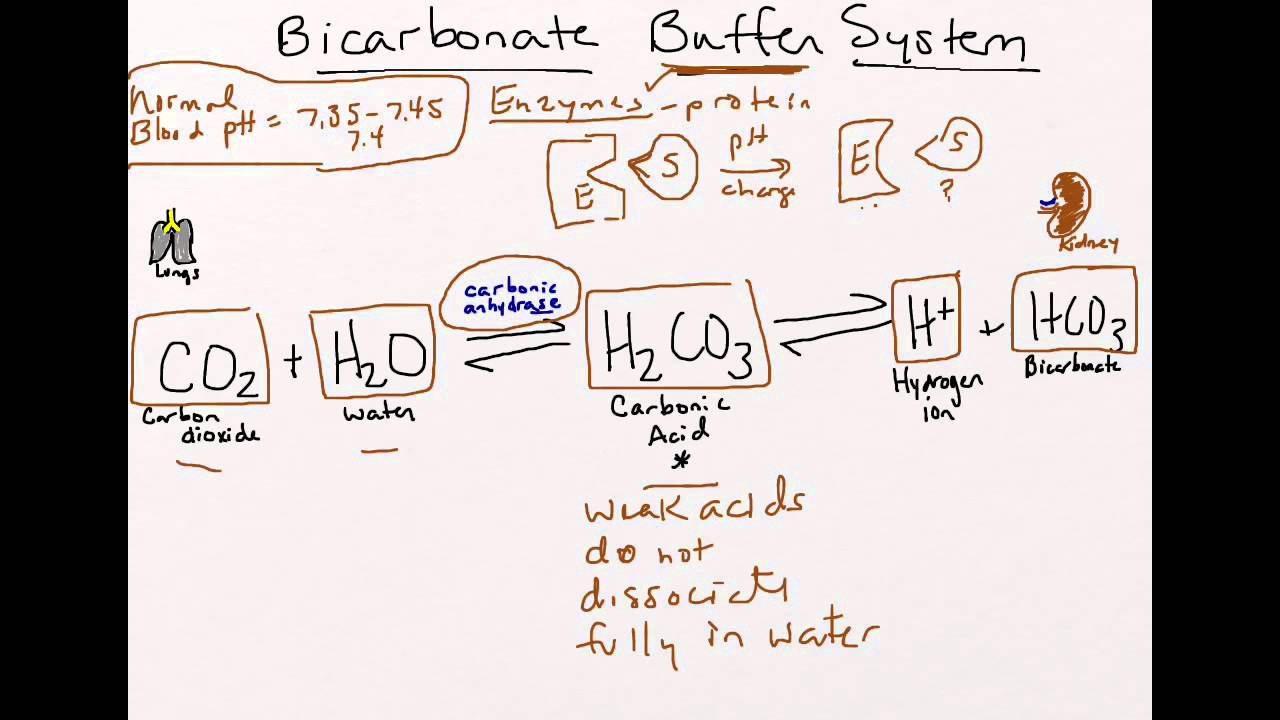
Bicarbonate Buffer System System Study Tips Pharmacy School

Smyle Plastic Free Toothpaste Only Natural Ingredients All Natural Toothpaste Toothpaste Natural Toothpaste

The Ultimate Guide On Food Grade Potassium Carbonate E Ingredients Com Carbonate Potassium Bicarbonate Food Additives

Caveman Chemistry Chemistry Education College Chemistry Chemistry Lessons

Pin By Pamela Griffiths On Alkaline Movement Potassium Bicarbonate Sodium Bicarbonate Alkaline Diet

Pin By Little Brianna On Laboratory Base Excess Actual Carbonate

Pin By Jennifer Clarke On Questions And Coriousty Things To Come Sodium Bicarbonate Pure Products

Seawater Carbonate Chemistry Ocean Acidification Carbonate Ocean Science

Ocean Acidification Needs Attention Livingseas Ocean Acidification Oceanography Lessons Oceanography

Chinese Snow Brand Sodium Bicarbonate Food Grade Sbc Baking Soda Sodium Hydrogen Carbonate Manufacturing Sodium Bicarbonate Baking Soda

Carbonate Bicarbonate Buffer Ph 9 2 To 10 6 Preparation Guide And Recipe Recipe Can Be Automatically Scaled By Entering D Preparation Recipes Chemistry Labs


Posting Komentar untuk "Carbonate To Bicarbonate"