Carbonate And Bicarbonate Buffer System
CO 2 is released from the blood increasing the blood pH right image. Carbonate buffers Theory A classic buffer is a combination of a weak acid and its conjugate salt.

Kordon 31261 Amquel Ammonia Detoxifier For Aquarium 1 Gallon Gallon Aquarium Water Treatment Water Treatment
Commonly used for various immunoassay applications and for many protein and antibody conjugation procedures including sandwich ELISA which require experimental surface coatings.

Carbonate and bicarbonate buffer system. The kidneys help control acid-base balance by excreting hydrogen ions and generating bicarbonate that helps maintain blood plasma pH within a normal range. Well in any buffer system the boost in. Sodium Carbonate Sodium Bicarbonate Buffer Catalog Number 14000020 Part of Kits.
When atmospheric carbon dioxide is dissolved in seawater carbonic acid H 2 CO 3 is formed. Carbonate that follows this path represents a linkage between the carbon cycle and the hydrologic cycle. This is responsible for about 80 of extracellular buffering.
One simple way to. Buffer Identification of Manufacturer. Using optical traps to manipulate single DNA strands.
Therefore at pH 85 you will have both carbonate and bicarbonate present. However in most H 2 CO 3 as defined below is used to represent solution carbonates. The carbonatecarbonic acid buffer the phosphate buffer and the buffering of plasma proteins.
However ecient transfer. While the third buffer is the most plentiful the first is usually considered the most important since it is coupled to the respiratory system. The bodys chemical buffer system consists of three individual buffers.
For instance carbonic acid H 2CO 3 and sodium bicarbonate NaHCO 3 or even sodium bicarbonate and calcium carbonate. The co-existence of these species in seawater creates a chemical buffer system regulating the pH and the pCO 2 of the oceans. Of course it all depends on what one wants to accomplish.
H 2C O 3 H 2 C 3 CO 2aq 2 Where H 2 CO 3. The major buffer system in the ECF is the CO 2-bicarbonate buffer system. Carbonate-Bicarbonate Buffer pH 92 to 106 preparation guide and recipe.
In the case of carbonatebicarbonate that would be an equi-molar ratio of carbonate and bicarbonate. When water is in equilibrium with both CO 2 form the atmosphere and carbonate containing rock the pH of the water is buffered to a pH of 83 close to the pKa of the weak acid bicarbonate HCO 3-pKa 84. In physiology the body uses the carbonic acidbicarbonate system to adjust the pH of blood.
Protein buffer systems work predominantly inside cells. They are in about a 101 ratio. Carbon dioxide plays a vital role in the chemistry of sea water.
CO 2 enters the blood decreasing the blood pH left image. CarbonateBicarbonate Buffer 92 Chemical Name. The carbon dioxide that is dissolved by naturally circulating waters appears in chemical analysis principally as bicarbonate and carbonate ions.
Importantly the weak acid form of the bicarbonate buffer H 2 CO 3 is rapidly inter-converted by carbonic anhydrase into. Of its transformation into bicarbonate and carbonate in a slightly alkaline aqueous medium and they contain about 60 times as much inorganic carbon as is in the atmosphere. When the first proton is donated HCO3- otherwise known.
CarbBicarb Buffer pH 92 01. This buffer functions in the same way as the phosphate buffer. Acids in acid rain promote the dissolution of calcium carbonate by reacting with the carbonate anion.
The pH of blood depends on the ratio of carbon dioxide to bicarbonate. The carbonatebicarbonate anion system has two pK a values one at 64 and one at 103. The carbonate system which is the major source of bufiering in the ocean and is the main subject of.
The important thing to realize here is that carbonic acid H_2CO_3 is actually formed when carbon dioxide CO_2 is. CO 2 aq OH- HCO 3 - 1 In biological systems the hydration of carbon dioxide is catalyzed by carbonic anhydrase a Zn-containing enzyme. In natural systems there are many buffers.
The blood buffering system is. Carbonic acid is diprotic which means in has two H ions to donate to solution. Recipe can be automatically scaled by entering desired final volume.
Additional H is consumed by HCO 3 and additional OH is consumed by H 2 CO 3The value of Ka for this equilibrium is 79 10-7 and the pKa is 61 at body temperatureIn blood plasma the concentration of hydrogen carbonate ion is about twenty times the concentration of carbonic. Carbonic acid H2CO3 a compound. In humans and other animals the carbonate buffering system helps maintain a constant pH in the bloodstream.
It is the most important ECF buffer for metabolic acids but it. Carbon Dioxide-Bicarbonate-Carbonate Equilibrium One important buffer in surface waters is the carbon dioxidebicarbonatecarbonate buffer. The buffer systems functioning in blood plasma include plasma proteins phosphate and bicarbonate and carbonic acid buffers.
How does a carbonate buffer system work. The Bicarbonate Buffer System. Other mechanisms that assist in this function include the hemoglobin molecule in your red blood cells which also helps to buffer blood pH.
The influence of the bicarbonate-carbon dioxide buffer system on the pH gradient delta pH across the inner membrane of mitochondria from rabbit renal cortex was studied with and without phosphate in the medium. The carbonic acid - bicarbonate buffer system consists of carbonic acid a weak acid and the bicarbonate anion its conjugate base. Carbonic acid is divalent.
Only a small portion of the aqueous carbon dioxide exists as carbonic acid. That is it can undergo two de-protonation reactions to form bicarbonate HCO 3- and carbonate CO 3 2-. Limestone is one familiar form of calcium carbonate.
The Carbonic AcidBicarbonate Buffer. The bicarbonate buffering system is an crucial buffer system in the acid-base homeostasis of all living things. The CarbonateBicarbonate Buffer System.
The main role of the bicarbonate system is to regulate and control the pH of blood and counteract any force that will alter the pH. Bicarbonate HCO 3- can reversibly bind a free hydrogen ion and in doing so acts as a buffer against pH changes Review Buffer BasicsThe pK of the bicarbonate buffer is 61 providing excellent buffering capacity around the normal ECF pH of 74. Product and Company Identification Product Identifier.
In the human stomach and duodenum the bicarbonate buffer system serves to both neutralize gastric acid and stabilize the intracellular pH of epithelial cells via the secretion of bicarbonate ion into the gastric mucosa. Calcium carbonate CaCO 3 is a very common mineral. What is the chemical equation for the carbonic acid buffer system.
For some purposes you might want a weaker buffer. Delta pH with bicarbonate buffer or phosphate in the medium was greater at low than at high medium pH so that the difference delta delta pH between delta pH. The role of the bicarbonate buffer system in regulating blood pH This is the currently selected item.
A buffer system exists to help neutralize the blood if excess hydrogen or hydroxide ions are produced. What happens when you titrate this combination with the strong acid of your choice. One that is important in surface waters is the carbonic acidbicarbonate buffer.

Tanganyika Buffer 4 Kg 8 8 Lbs Pet Supplements Fish Pet Koi Fish Food

Hagen Rim Clamp For Aquaclear 201 301 Powerhead Rim Aquarium Supplies Clamp

10 99 10 07 For Coral Propagation Coral Accel Should Be Used Daily It Is A Clean Product However Water Quality Should Always Be Soft Corals Marine Coral

Aussie Symphyllia Wysiwyg Live Coral Ebay In 2021 Live Coral Underwater Creatures Coral

Esv Aquatics Bionic 2 Part Calcium Buffer System For Aquarium Healthy Digestive System Healthy Fish Koi Fish Food

Liquid Marine Buffer 20 L 5 3 Fl Gal Aquarium Water Treatments Marine Marine Aquarium

Seachem Reef Buffer 8 8 Fl Oz Petco In 2021 Saltwater Tank Reef Aquarium Planted Aquarium

Pin On Fish Aquarium Water Treatments

Reef Calcium Is A Concentrated 50 000 Mg L Bioavailable Polygluconate Complexed Calcium Intended To Maintain Calcium In T Calcium Amazon Deals Shopping Reef

Chemistry Investigatory Project On Antacids Investigatory Project Chemistry Chemistry Projects
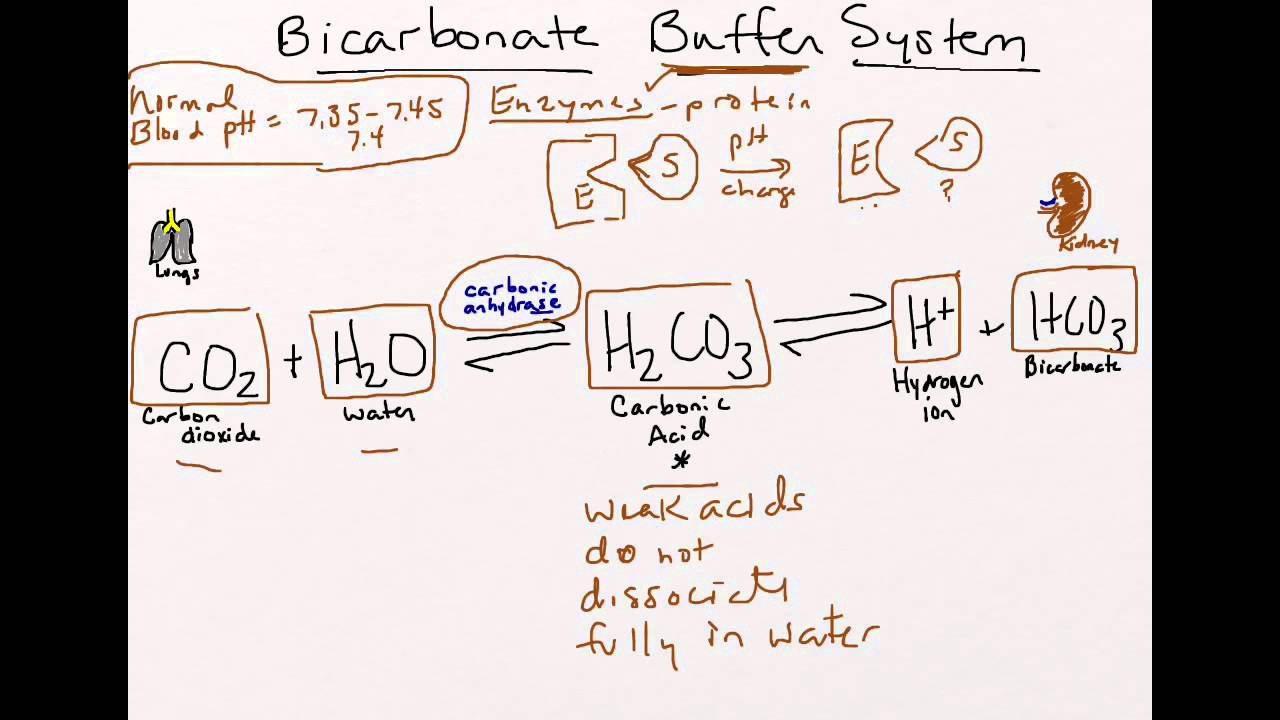
Bicarbonate Buffer System System Study Tips Pharmacy School



Posting Komentar untuk "Carbonate And Bicarbonate Buffer System"